 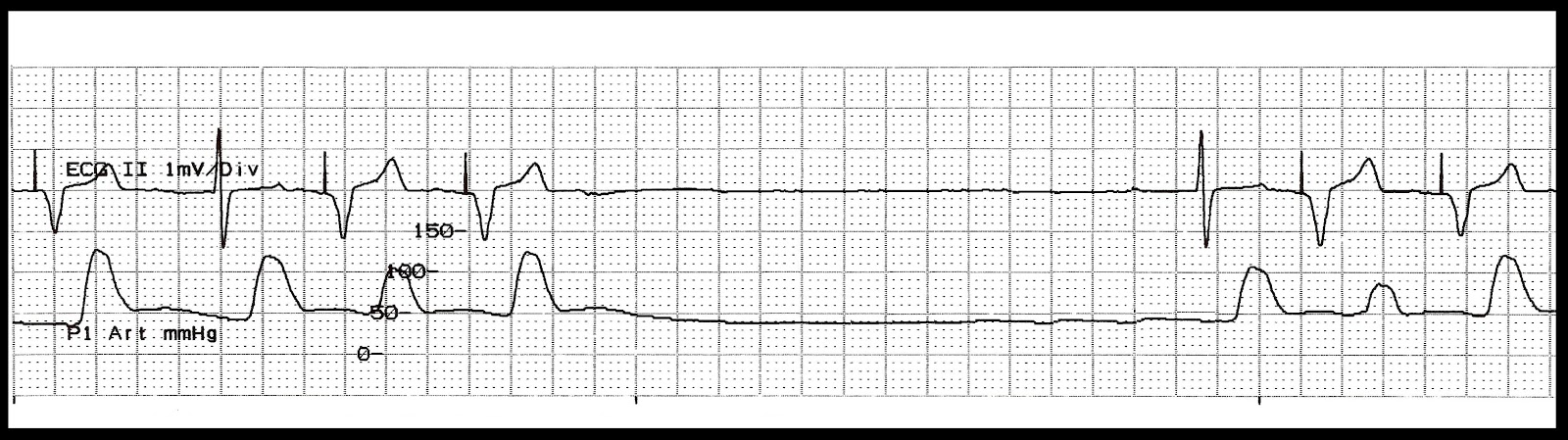
(See Pacemaker comparison.) The device is 25.9 mm long, 6.7 mm wide, and weighs 2 grams. Medtronic won Food and Drug Administration (FDA) approval to market the Micra LP on April 6, 2016. In the United States, just 20% of patients who required permanent pacemakers received traditional single-chamber devices, while the remaining 80% received traditional dual-chamber devices. The 2009 world survey by Mond and Proclemer provides prevalence data on the use of pacemakers by type. LPs are single-chamber devices, so currently they’re available only for a limited number of pacemaker candidates. LPs, which are miniature devices, are implanted directly into the right ventricle via the femoral vein in the groin. The LP was developed to avoid the complications associated with the leads and surgical pocket required when placing a traditional pacemaker. Understanding this new technology will help you deliver safe care to patients who may be candidates for an LP or who may already have one. Many nurses may not have encountered a patient with an LP and little nursing literature on the topic exists. (See Traditional pacemaker basics.) However, as with any new technology, a knowledge gap exists. This device is one of the biggest advancements in pacemakers since they came into use for symptomatic bradycardia more than 50 years ago. 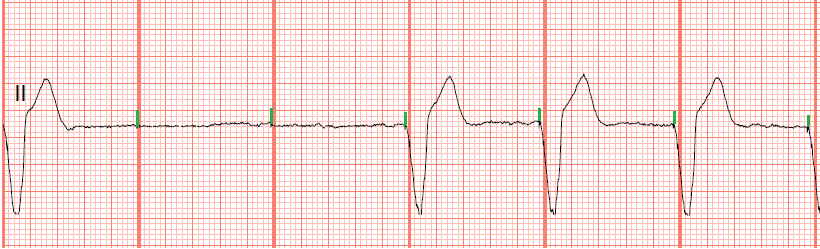
ONE of the most recent developments in cardiac pacing is the leadless pacemaker (LP).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
